Interferon-beta in MS
Quite contrary to the other therapy sections, this is no review. The
reason for bringing this page is simply that I want to make the figure
accessible; and the reason for not making a review is that this has been done
already, that treatment with Interferon-beta [IFNB] does not
represent an alternative treatment but currently one of the few (probably the
best) attempts to slower progression of the disease, and finally that the big
labour associated with a literature survey is almost bound not to alter the
before mentioned statement, although it might establish its background further
See addition below concerning neutralizing antibodies.
.
It is my impression
that
1. IFNB in
the higher dosage (28-32 MIO units per week) produces the best results, as
compared to the lower dosage of 6 MIO in a single weekly injection;
2. there is no therapeutic difference in activity between IFNB
1a and 1b;
3. cross-reaction makes a change of therapy under the
impression of antibodies useless;
4. immunosuppressive therapy is inferior, but occasionally
required in addition to IFNB; and
5. copolymer-A (glatimer racemate) and intravenous immunoglobulines,
while not being well-defined drugs, exerts only an unspecified (irritative) immune response which makes them incomparable
(possibly inferior) to IFNB.
6.
IFN-Gamma has often been shown directly damaging in MS, IFN-alpha, while it has
not been directly compared with IFNB, has some effect but is probably acting
less convincing. Some hope is now carried on the newly synthetized
IFN-tau - the future may show
if it works.
It would
certainly have looked more impressively if this was the conclusion of a large
review: It is not, and you will only seldom find so sharply defined statements,
for that reason we leave it as an impression.
With the
injection of IFNB, the patient should try to find a new place each time, so
that a new injection in the same area nay only take place after a certain span
of time. Using the indication of the figure below, 1-2 months will pass before
an area is approached again. Of course, other areas might be defined instead,
it is only important that a schedule is made.
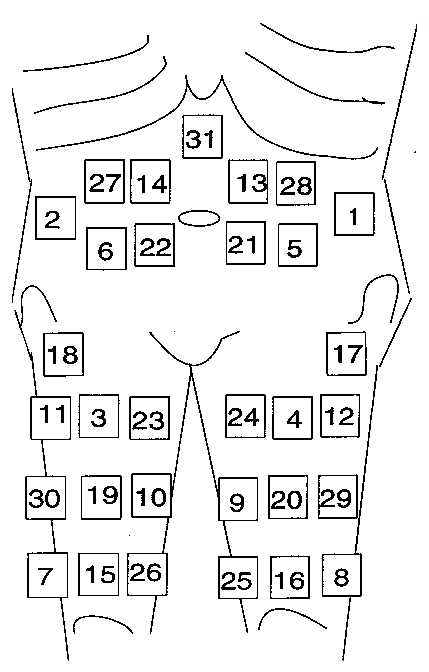
Figure:
Daily injection sites for IFNB.
Addition:
A rare study form desires reference, partly for its conclusion, partly
while it of the type I prefer (studying what happens to
different patients) and not the unethical randomized placebo-controlled trials
commented upon elsewhere (A Conflict to Come, Placebomania in MS-Studies, MS-Science
2004).
The Danish National IFN-beta Project [1] is the
largest independent study that has examined the impact of neutralizing
antibodies [NAbs] on the therapeutic effect of IFNB. The study comprised
541 patients, who all were treated with the same IFN[beta] preparation throughout the entire observation
period, beginning during the years 1996 to 1999 in
NAbs were found
in 45,4% of 194 patients receiving IFNB-1B and 45,7%, the same incidence, in 162
patients receiving IFNB-1A three times weekly. In contrast, only 14.6 % of 185
patients, receiving IFNB-1A developed NAbs. This
finding must, however, be balanced upon a lessened activity, assumed from the
lower dosage (22 or 30 µg weekly). The net effect upon the EDSS was not disclosed
in this study.
The status at 12 months was somewhat predictive:
if the 12-month status was negative, only 9%, 9%, 12%, and 15% became NAb positive after 2, 3, 4, and 5 years, respectively.
Conversely, if the 12-month status was positive, the percentages of these
patients who were still NAb positive after 2, 3, 4,
and 5 years were 82%, 69%, 53%, and 46%, respectively
The name “neutralizing” is well selected, also what the
effect concerns. The relapse-rate was 55% higher during NAb positive periods as
compared with NAb negative ones. Again, this
apparently clear statement is relativised through the
chances for the NAb positive patient at 12 months to
become NAb negative, which appear to be even after
about 4.5 years.
I hope that other conclusions may be published from this exceptional
investigation. Moreover, I hope that more will be published from such kind of
studies, investigating various habits of thousands of patients over the years.
But then, there are the double-blinded editors …
1. Sorensen PS, Ross C, Clemmesen KM, Bendtzen K, Frederiksen JL, Jensen K, Kristensen O, Petersen T, Rasmussen S, Ravnborg M, Stenager E, Koch-Henriksen N; Danish Multiple Sclerosis Study Group; Danish Interferon-beta Project. Antibodies to IFN-beta: the Danish National IFN-beta Project. Neurology 2003;61(Suppl 5):S27-8.
Written